Research
The long-term interest of my laboratory is to describe why there is an apparent dichotomy in the regulation of gastrointestinal performance among ectotherms and variation in metabolic and tissue performance, and how tissues are able to respond both functionally and structurally to changes in demand. To address the "why" questions, we undertake comparative studies using fishes, amphibians, and reptiles to examine the correlation between natural feeding habits and the regulation of gastrointestinal performance and metabolic rate. To explore the "how" questions, we search for the signals that trigger tissue responses and the mechanisms that underlie those responses. For this we rely primarily on our model system, the Burmese python, and employ an array of methods to describe the signaling, cellular, and molecular mechanisms of tissue plasticity. The following briefly describes the major studies being undertaken in my laboratory. There are additional related projects being undertaken in collaboration with other researchers and being conducted by my students.
Evolution of the adaptive interplay between feeding ecology and digestive physiology:
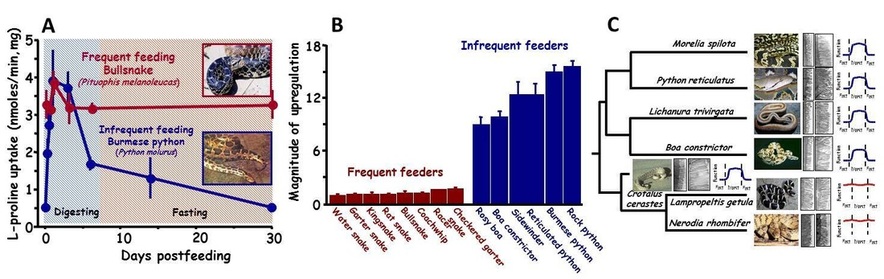
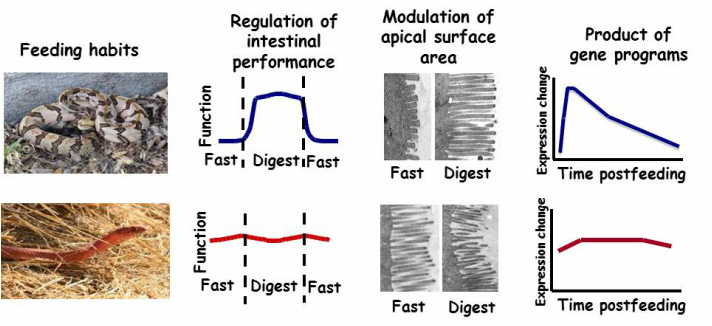
This long-term program explores the adaptive interplay between feeding ecology and digestive physiology with a focus on fishes, amphibians and reptiles. This work has demonstrated the link between feeding frequency and the magnitude by which digestive performance is regulated. Species that feed relatively frequently in the wild regulate their intestinal performance very modestly, whereas those species that naturally experience long intervals between meals regulate their intestinal performance over much greater ranges. To our knowledge, the capacity to widely regulate intestinal performance has evolved independently at least five times among reptiles and amphibians that naturally experience long episodes of fasting. We have hypothesized that the selective pressure underlying the evolution of these two modes of gastrointestinal responses resides for both in the conservation of energy. The cellular mechanisms that underlie each of these different modes of intestinal response is the extent that the microvilli are altered with feeding and fasting. We have found for animals that narrowly alter intestinal function that they experience little change in microvillus height with feeding and fasting. However, species that widely regulate intestinal performance do so by dramatically shortening their microvilli with fasting and then rapidly lengthen them with feeding. For both modes, the modulation in functional surface area determines the degree of intestinal regulation. We are now exploring whether modes of intestinal regulation are constrained by phylogeny (i.e., conserved within a clade regardless of feeding habits) or have co-evolved independently with feeding habits within clades.
(A) Postfeeding profiles of intestinal L-proline uptake for the frequently feeding bullsnake and infrequently feeding Burmese python. Illustrated is the modest regulation of intestinal function with feeding and fasting for the bullsnake and much greater regulation of intestinal function for the python. (B) Relative magnitude of upregulation of intestinal performance between frequently feeding and infrequently feeding snakes. On average frequently feeding snakes can at best double intestinal performance with feeding, whereas infrequently feeding species increase intestinal performance by 10-fold. (C) Phylogenetic representation of changes in microvillus length with feeding and the magnitude of intestinal response. Pythons, boids, and rattlesnakes experience 5-fold lengthening of their microvilli with feeding with similar increase in intestinal function, whereas colubrid snakes do not vary their microvillus length with feeding and thus experience no significant postfeeding change in function.
Evolution of the molecular mechanisms of intestinal response.
We are developing a program to explore whether independent evolved modes of intestinal response reflect shared sets of co-evolved gene expression changes. Given the similarities in the phenotypic responses among snakes that feed infrequently (or frequently), we hypothesize that these snakes share similar gene expression programs that have been co-opted across lineages. An alternative hypothesis is that similar phenotypic responses among lineages stem from different suites of expressed genes that have each arisen independently with that mode of intestinal response.
Sit-and-wait and active foraging feeding habits have co-evolved with distinctly different modes of intestinal regulation with feeding and fasting, a function of differences in the remodeling of the intestinal microvilli, which is a product of two different gene expression programs.
Determinants of metabolic rate and digestive energetics
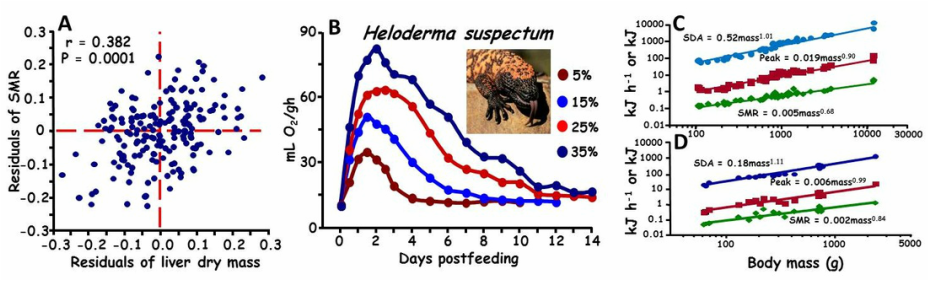
We have on-going projects that examine the determinants of metabolic rate and the sources for intraspecific and interspecific variation in metabolic rates. We examine hypotheses on how differences in organ masses, body composition, and body size are responsible for variation in metabolic rates. We also conduct numerous studies exploring the effects of meal size, meal type, meal composition, body temperature, and body size on postfeeding metabolism and specific dynamic action (SDA). SDA is the cumulative energy expended on all processes of meal digestion and assimilation, and is highly dependent upon characteristics of the meal and body.
(A) Residual plot illustrating that individual diamondback water snakes with larger than predicted liver also possess greater than predicted standard metabolic rates (SMR). (B) Postfeeding profiles of metabolic rate of the Gila monster at four different meal sizes (5, 15, 25, & 35% of body mass). Illustrated is that with an increase in meal size, there is an increase in the metabolic response and duration of elevated metabolism. (C). Intraspecific scaling for the Burmese python of SMR, peak postfeeding metabolism, and specific dynamic action (SDA). (D) Interspecific scaling for snakes of SMR, peak postfeeding metabolism, and SDA. SMR scales allometrically, whereas SDA scales isometrically with body mass.
Regulatory mechanisms of phenotypic flexibility
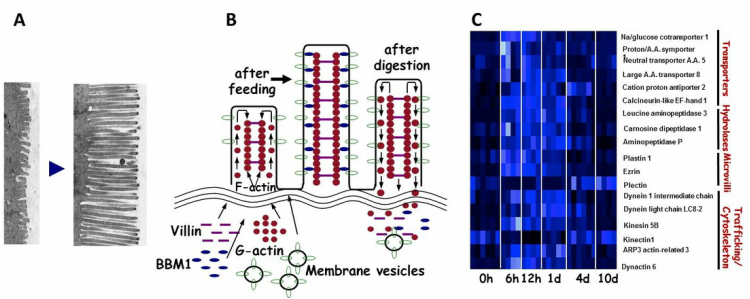
Infrequently feeding snakes (notably pythons and boas) experience with feeding 35-100% increase in the mass of the heart, liver, small intestine, pancreas and kidneys, 5 to 10-fold increases in specific activities of the pancreas and small intestine, and a massive increase in gastric acid production. Upon the completion of meal digestion, tissue experience atrophy, function is down regulated, and the stomach ceases acid production. These snakes exhibit extreme repeated flexibility of tissue structure and performance. We are therefore using these snakes to seek out the signals that trigger such rapid tissue response and cellular and molecular mechanisms that underlie the unprecedented modulation of tissue and form and function. This program is extremely integrated and employs techniques of histology, immunohistochemistry, proteomics, and transcriptomics.
(A) Posfeeding lengthening of the Burmese python intestinal micorvilli. (B). Proposed mechanism of microvilli lengthening with feeding and shortening upon the completion of digestion involving the trafficking microvillus proteins. (C). Heat map illustrating changes in the expression of genes involved in the remodeling of the python small intestine with feeding.
Translation of signaling and regulatory factors to mammalian systems.
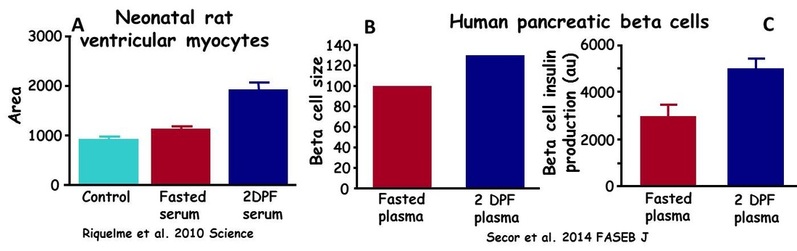
We have already demonstrated that plasma from a fed python can stimulate hypertrophy of cardiomyocytes of juvenile rats. We are currently exploring studies on whether small molecules in the blood of digesting python can similarly stimulate the growth and increase performance of other cells (e.g., pancreatic beta-cells) and tissues of mammals. A potential application is the use of such identified active molecules to enhance the performance of human tissues that have been suppressed by surgery and disease (e.g., diabetes).
(A) Administration of serum from fasted Burmese pythons had no effect on the size of neonatal rat cardiomyocytes, whereas serum from a digesting snake stimulated a 70% in the size of the rat's cardiomyocytes. (B). Plasma from a digesting Burmese pythons (2 days postfeeding) induced a significant increase in the size of human pancreatic beta cells. (C) Human pancreatic beta cell when exposed to plasma from a digesting pythons experience a 60% increase in insulin production.